
Material Harvest® Reverse Phase Silica Gels
| Product Name | Reverse Phase C18 Silica Gel |
| Synonym | Octadecyl Bonded Silica Gel |
| Loading | 17% carbon content |
| Particle Size | 40 – 63 microns (mesh 230 – 400) |
| Average Pore Size | 60 Å |
| Mean Surface Area | 500 m2/g |
| Pore Volume | 0.8 ml/g |
Reverse phase silica gel is used in organic chemistry for the purification and isolation of synthetic compounds (i.e. post-reaction “harvesting” of chemical products).
Our reverse phase silica gel is manufactured in accordance to the highest specifications. As shown in Figure 1, the synthetic process entails grafting octadecyl moieties on the surface of regular silica gel, followed by end-capping of the residual OH groups. As as result of this end-capping, C18 silica gel is insoluble in polar solvents including methanol.
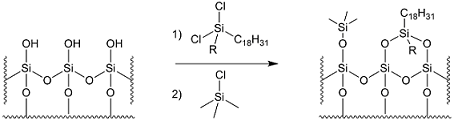
Figure 1 – Surface functionalisation of regular silica gel, followed by end-capping
The most common grade of C18 reverse phase silica is based on silica gel 60, and has a particle size distribution of mesh 230 – 400 (i.e. 40 – 63 µm). The number "60" in "Silica Gel 60" means that the pore diameter of the silica particles is 60 Å.
In terms of the loading or carbon content, 17% is the most common configuration. Reverse phases having "high" (23%) or "low" (11%) carbon content are also available on demand.
Product Code |
Specifications |
Pricing
|
|||
100 g |
500 g |
1 kg |
20 kg |
||
MH 0500601 |
mesh 230 – 400; 60 Å; 17% loading |
£152.00 | £627.00 |
£1,118.00 | contact us |
We offer academic and volume-based discounts - please contact us for details.
Column Chromatography with Exotic Reverse Phases
Other reverse phases, for example C8, C4, C1, C-adamantane, C-cyclehexane, C-phenyl and C-cyano are also available. For detailed information on these exotic stationary phases please refer to our dedicated page on functionalised silica gels.
These stationary phases exhibit unique separation parameters that allow specific molecules to be isolated. For example, large biomolecules with extensive hydrophobic regions may not be amenable to purification using C18 silica gel - in these cases, C4 or C1 silica gels may be more appropriate to use.
If you wish to test out a variety of reverse phase silica gels, so as to find the most ideal sorbent for a specific separation, please refer to the "reverse phase kits" below. These kits are compilations of reverse phases packed together in a single box-set.
Product Code |
Each kit contains multiple reverse phases including C18 varieties (23%, 17% etc), and other sorbents such as C12; C8; C4; C-phenyl etc |
||||
5 g of each |
10 g of each |
25 g of each |
50 g of each |
100 g of each
|
|
Reversed-Phase kit |
|||||
Lecture Notes on Reverse Phase Column Chromatography
Column chromatography is a purification technique used in organic chemistry and synthesis. It can potentially isolate a compound from a mixture of unwanted substances – for example to separate the product of a chemical reaction from its crude mixture.
Column chromatography utilises a stationary phase, otherwise known as a sorbent or an adsorbent, which is normally packed in a glass column. Common examples of stationary phases include silica gel and alumina. The mixture of compounds is applied at the top of the column and a solvent or solvent mixture, known as the “eluent” or “mobile phase”, is allowed to flow through the silica gel or alumina.
In reverse phase column chromatography, the stationary phase is functionalised with an alkyl substituent such as octadecyl – an alkyl chain having 18 carbon atoms. This type of silica is commonly referred to as C18 silica; it is the most common reverse phase available in the market. Other reverse phases include C8, C4, C1, C-cyclohexyl, C-phenyl and C-cyano.
Polar compounds elute faster than their non-polar counterparts when reverse phases are employed. This is the opposite situation in respect to chromatography with unmodified or "regular" silica gel, therefore the technique is traditionally referred to as being "reversed".
The degree of hydrophobicity decreases as the carbon chain length also decreases: in other words, C18 has the highest degree of hydrophobicity and C1 has the lowest degree of hydrophobicity. It therefore follows that shorter chain lengths like C1 and C4 are better where less retention towards non-polar compounds is needed. Thus, if your compounds elute very slowly over C18 silica gel, an alternative reverse phase such as C4 may be more useful.
If you would like to receive more information please contact us.
![]()
![]()
 |